Making
Microorganisms Mobilize Soil Phosphorus
Alan E.
Richardson
CSIRO Plant Industry, PO Box 1600, Canberra ACT 2601, AUSTRALIA
alan.richardson@csiro.au
__________________
Microorganisms are involved in a range of
processes that affect the transformation of soil phosphorus (P) and are thus an
integral component of the soil P cycle.
In particular, soil microorganisms are effective in releasing P from
inorganic and organic pools of total soil P through solubilization and
mineralization. The microbial biomass
in soil also contains a significant quantity of immobilized P that is
potentially available to plants.
Microorganisms therefore are critical for the transfer of P from poorly
available soil pools to plant available forms and are important for maintaining
P in readily available pools. These processes are likely to be most significant
in the rhizosphere of plants.
Consequently, there has been longstanding
interest in the manipulation of soil microorganisms to improve the P nutrition
of plants, with the objective of increasing the overall efficiency of P-use in
agricultural systems. This
interest stems from the fact that P deficiency is widespread on soils
throughout the world, that P fertilizer represents a major cost for
agricultural production and that the efficiency of P-use by plants from soil
and fertilizer sources is poor.
Furthermore, P is a finite resource. Based on current rate of use, it is expected that the
worlds’ known reserves of high quality rock phosphate will be depleted
within the current century (Isherwood, 2000). Beyond this time the production of phosphate-based
fertilizers will require the processing of lower-grade rock phosphates at
significantly higher cost.
Alternatively, the direct use of rock phosphates as fertilizers will require
an effective means for solubilization. These issues are particularly relevant
to soils throughout developing countries and on acidic soils in tropical and
subtropical regions (Hedley et al., 1995). It is also imperative that management of P
fertilizers in agricultural environments is improved (particularly in more
highly P fertilized environments) so that any adverse environmental effects due
to P losses are minimized (Tunney et al., 1997).
The concept of using soil microorganisms to
improve mobilization of poorly available forms of soil P is not new. It is now
some 50 years since Gerretsen (1948) first showed that pure cultures of soil
bacteria could increase the P nutrition of plants through increased solubility
of Ca-phosphates. Volumes of
literature have since been published, a great deal has been promised, but it is
fair to say that not much has been delivered. Clearly, microbial-plant interactions in soil environments
are complex and, with few exceptions, have proven difficult to manipulate (reviewed
by Richardson, 2001). Therefore,
the challenge remains. Indeed,
opportunities for exploiting soil microorganisms for P-mobilisation are
improved as knowledge of the processes and understanding of the ecology of
microorganisms in soil environments is gained. Such opportunities are further enhanced with the advent of
new techniques. These include the
possibility for direct manipulation of organisms through gene technology.
In this paper, recent issues concerning the
mobilization of soil P by microorganisms are summarized and some opportunities
for the future are discussed.
Phosphorus
mobilization by soil microorganisms
Microorganisms directly affect the ability
of plants to acquire P from soil through a number of structural or process-mediated
mechanisms. These include (i) an increase in the surface area of roots by
either an extension of existing root systems (eg, mycorrhizal associations) or
by enhancement of root branching and root hair development (ie, growth
stimulation through phytohormones), (ii) by displacement of sorption equilibria
that results in increased net transfer of phosphate ions into soil solution or
an increase in the mobility of organic forms of P and (iii) through stimulation
of metabolic processes that are effective in directly solubilizing and
mineralizing P from poorly available forms of inorganic and organic P. These processes include the excretion
of hydrogen ions, the release of organic acids, the production of siderophores
and the production of phosphatase enzymes that are able to hydrolyse soil
organic P (Figure 1). In
particular, organic acids and associated protons are effective in solubilizing
precipitated forms of soil P (eg, Fe- and Al-P in acid soils, Ca-P in alkaline
soils), chelating metal ions that may be associated with complexed forms of P
or may facilitate the release of adsorbed P through ligand exchange reactions
(Jones, 1998).
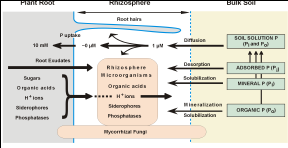
Figure 1. Schematic representation of major
physiological factors associated with plant roots and soil microorganisms that
influence the availability of soil P in the rhizosphere (from Richardson,
2001).
However, distinction between the roles of microbial
processes, as distinct from direct effects of plant mechanisms, on P
mobilization in soil is poorly understood. It is well established that plant roots effectively increase
P acquisition through modified root growth and architecture and similarly
produce metabolites that directly influence P availability (Raghothama,
1999). Processes such as
rhizosphere acidification, exudation of organic acids and secretion of
phosphatases from plant roots occur in response to P deficiency, and are
established mechanisms by which plants acquire P (Randall et al., 2001). Furthermore, it has been suggested that microbial-mediated
processes on their own may be insignificant in soil environments, and are
unlikely to mobilize sufficient P for plant requirements (Tinker 1980). This argument remains to be
resolved. On the other hand, the
importance of the microbial biomass for P cycling in soil and the potential of
this P to contribute to plant P nutrition is more difficult to deny.
Soil
microbial biomass phosphorus and contribution to plant nutrition
The microbial biomass in soil contains a
significant amount of P (typically 10 to 50 kg P/ha, but as high as 100 kg
P/ha) and generally accounts for 2 to 5% of the total P and around 10 to 15% of
the soil organic P. Importantly,
microbial P is a dynamic component of the soil P cycle and is responsive to
soil fertility, seasonal conditions and management practices. Whilst the P content of microbial
biomass may vary considerably in relation to microbial C, it is evident that
significant pools are maintained even in soils considered to be P deficient for
plant growth (Oberson et al., 2001). This indicates that microorganisms in soil are highly
efficient in acquiring P to meet their own requirements. In addition, it has been shown that
soil microorganisms are capable of rapidly assimilating P supplied from
fertilizer or as plant residues.
For instance, McLaughlin et al (1988) showed that some 25% of P in labelled crop
residues was incorporated into microbial biomass within 7 days.
A number of studies have highlighted the
potential importance of microbial P in providing available P to plants. Seasonal dynamics indicate that
significant amounts of P are released from the biomass in response to soil
moisture deficiency and it is estimated that soil microbial P is completely
turned over at least annually (He et al., 1997).
More recent studies have found that the rate of P-flux through the
microbial biomass is, in fact, considerably greater (Odel et al., 2000, Oberson et al., 2001). Incubation studies using labelled phosphate have shown both
a rapid incorporation of P into biomass (within 2 to 3 days) and concomitant
release of the P back to soil solution.
Significantly, these transfers of P occurred in the absence of any
significant changes in the size of the microbial P pool. Highest rates of P cycling through the
biomass were evident in P-deficient soil and in soils that received organic
inputs, as distinct from those that were P-fertilized. The capacity of the microbial
biomass to immobilize P was also increased by the provision of soluble C, which
resulted in an increase in both the size of the microbial P pool and its rate
of turnover.
These observations have important
implications concerning the contribution of microbial P to plant
nutrition. First, the significance
of P immobilization within the soil microflora and its effect on the
‘short-term’ availability of P to plants is not clear. Likewise, processes that affect the
release of P from the microbial biomass and its subsequent availability to
plants require further investigation.
Although P in microorganisms occurs predominantly in organic forms (or
as polyphosphates), the P appears to be rapidly mineralised and is readily
available for uptake by plant roots (Macklon et al., 1997). However, in soil environments the availability of released P
will be influenced by spatial and temporal factors and will also be subject to
further immobilization (by both soil micro- and macro-flora and fauna) and
other physico-chemicals reactions of P in soil. The actual contribution that P-turnover through
microorganisms makes to the mobilization of soil P therefore remains to be
fully determined. Nevertheless, it
is well known that soil P is significantly depleted in close proximity to
roots, that roots release significant quantities of C that is available to soil
microorganisms and that microbial populations in the rhizosphere are enhanced
by many orders of magnitude (Bowen and Rovira, 1999). Therefore, the potential for turnover of P by rhizosphere
microorganisms is substantial, and further work needs to be undertaken to
quantify it in terms of enhancing plant P nutrition.
Using
microorganisms to improve soil phosphorus availability
Recognition that microorganisms are
important for P mobilization in soil has led to research effort directed at
improving plant P nutrition. Essentially, there are two major strategies for
manipulating soil microorganisms:
(i) Management of existing microbial
populations to optimize their capacity to mobilize P.
Success with this approach requires
detailed knowledge of how soil management practices (eg, crop rotations, soil
amendments, cultivation, etc.) impact on microbial abundance, diversity and
presence of various functional groups and how these relate to the magnitude and
availability of different soil P fractions. The manipulation of VA mycorrhizas in soil through crop
rotation is one example of how populations might be managed to increase the
availability of soil P to plants (Thompson 1994). Increased mineralisation of organic P generally occurs in
response to soil cultivation and crop rotation has been shown to increase the
rate of P cycling through the microbial biomass. For example, incorporation of organic residues through
legume rotation resulted in higher biological activity and increased microbial
P uptake and release (Oberson et al., 2001).
Although the contribution of P released through these processes needs to
be evaluated in relation to plant uptake, such observations indicate that
management opportunities do exist for increasing the cycling of P and its
maintenance in plant-available pools. Elucidation as to whether or not the availability of
this P can be synchronized with plant requirements, or be targeted to the rhizosphere,
remains a significant challenge.
(ii) The use of specific microbial inoculants
to increase P mobilization.
A range of soil microorganisms able to
solubilize precipitated forms of P or mineralize organic P has been
characterized. Typically, such
organisms have been isolated using cultural procedures, with species of Pseudomonas and Bacillus bacteria and Aspergillus and Penicillium fungi being predominant. These organisms are commonly associated
with the rhizosphere and, when inoculated onto plants, often result in improved
growth and P nutrition with responses being observed under both glasshouse and
field conditions (see reviews by Kucey et al., 1989; Rodríguez and Fraga, 1999; Whitelaw,
2000). Despite this, there are few
examples of successful application of microbial inoculants. Essentially, a lack of consistent
performance under different environmental conditions in the field has precluded
their wider use. A number of
factors can be identified to explain this variable performance (Richardson,
2001). They include (i) poor
understanding of the actual mechanisms involved in plant growth promotion
where, in fact, P mobilization may not necessarily be the primary mechanism
involved, (ii) selection of microorganisms by laboratory screening may be
insufficiently rigorous when organisms are required to mobilize P in soil
environments, (iii) the apparent lack of any specific association between
phosphate solubilizing microorganisms and host plants, (iv) poor understanding
of interactions between physical and chemical characteristics of soil and how
these interact with biological P availability, (v) poor knowledge of how to
deliver microorganisms into soil environments and of how to establish them as
dominant components of complex microbial communities and, in particular, of
their capability of colonizing the rhizosphere, and (vi) in most instances the
benefits of microbial mobilization of P may in fact be indirect. In short, whilst microorganisms may
directly solubilize P to meet their own requirements, subsequent benefits to
plants may only occur following turnover of the microbial biomass.
It is evident therefore that the
proposition for developing inoculants for routine application remains
problematic and a number of issues still need to be addressed. Nevertheless, some microorganisms show
consistent plant growth promotion under glasshouse and field conditions and
have been developed as commercial inoculants (eg., Penicillium spp., Leggett et al., 2001). Although growth promotion in such cases is generally
associated with increased plant P nutrition, it is difficult in many cases to
ascertain whether increased P-mobilization is either the cause or the
consequence of the response.
Similarly, many other organisms (including mixed populations of soil bacteria
and fungi, that often are only poorly characterized) have been promoted as
commercial inoculants with claims that they increase plant growth through
P-mobilization. Unfortunately, in
most cases detailed evidence to support these claims does not exist.
Prospects
for enhancing phosphorus mobilization by soil microorganisms
There seems little doubt that soil
microorganisms are essential for the cycling of P in terrestrial ecosystems and
as such, play an important role either directly or indirectly in mediating
phosphate availability to plants.
However, attempts to capitalise on microbial processes to increase plant
access to P from soil and/or fertilizer sources have generally met with limited
success. It is reasonable to
assume that future opportunities will increase as understanding of the
processes of P mobilization and the ecology of microorganisms in soil
environments improves. Development
of novel techniques and access to new technologies will be important. Recent developments in microbial
community analysis that do not rely on cultural procedures will provide better
understanding of how microorganisms interact in complex environments. For example, elegant procedures are now
available for detection and visualization of specific microorganisms in the
rhizosphere (eg, Ranjard et al., 2000, Theron and Cloete, 2000).
Opportunity also exists for genetic
manipulation of soil microorganisms. It is now a reality that gene technologies can be used to
enhance specific traits that may increase an organism’s capacity to
mobilize soil P directly, enhance its ability to colonize the rhizosphere (ie,
rhizosphere competence, Lugtenberg et al., 2001), or even to form specific associations with
plant roots (Bowen and Rovia, 1999).
Alternatively, microorganisms may provide a novel source of genes for
directly modifying plants. For
example, it has been reported that, when expressed in roots, a bacterial citrate
synthase gene increases the exudation of organic acids and significantly
improves plant access to soil P (López-Bucio et al., 2000). The credibility of this report has however been questioned
(Delhaize et al.,
2001). The ability of plants to
use P from phytate, which is a predominant form of organic P in most soils, has
been shown to be dependent on the presence of soil microorganisms and
utilisation of phytate-P was significantly improved when an Aspergillus phytase gene is expressed directly in plant
roots (Richardson et al.,
2001a, Richardson et al.,
2001b). Although such approaches
require further validation, particularly in soil environments, they highlight
that new opportunities do exist.
However, it is also important to recognize that any approach to increase
the mobilization of soil P through genetic modification, whether it be of soil
microorganisms or plants themselves, will also need to satisfy a range of
community issues.
The promise of exploiting soil
microorganisms to increase mobilization of soil P remains. Whether or not this will be achieved
through better management of soil microbial communities, by development of more
effective microbial inoculants, through the genetic manipulation of specific
organisms or with a combination of these approaches is not known. Nevertheless, what is clear is that
soil microorganisms play an important role in the mobilization of soil P and
that detailed understanding of their contribution to the cycling of P in
soil-plant systems is required for the development of sustainable agriculture.
REFERENCES
Bowen G.D. and Rovira A.D. (1999) The rhizosphere and its
management to improve plant growth. Advances in Agronomy. 66, 1-102.
Delhaize E., Hebb D.M. and Ryan P.R. (2001)
Expression of a Pseudomonas aeruginosa citrate synthase gene in tobacco is not associated
with either citrate accumulation or efflux. Plant Physiology 125, 2059-2067.
Gerretsen F.C. (1948) The influence of
microorganisms on the phosphate intake by the plant. Plant and Soil 1, 51-81.
He Z.L., Wu J., O’Donnell A.G. and
Syers J.K. (1997) Seasonal
responses in microbial biomass carbon, phosphorus and sulphur in soils under
pasture. Biology and Fertility
of Soils 24, 421-428.
Hedley M.J., Mortvedt J.J., Bolan N.S. and
Syers J.K. (1995). Phosphorus
fertility management in agroecosystems. In Phosphorus in the global
environment: Transfers, cycles and management. (Ed H. Tiessen), pp. 59-92, Wiley & Sons.
Isherwood K.F. (2000) Mineral fertilizer
use and the environment.
International Fertilizer Industry Association / United Nations Environment
Programme, Paris.
Jones D.L. (1998)
Organic acids in the rhizosphere - a critical review. Plant and Soil 205, 25-44.
Kucey R.M.N., Janzen H.H. and Leggett M.E.
(1989) Microbially mediated increases in plant-available phosphorus.
Advances in Agronomy 42, 199-228.
Leggett M., Gleddie S. and Holloway G.
(2001). Phosphate-solubilising microorganisms and their use. In Plant
nutrient acquisition: New perspectives (Eds N. Ae, J. Arihara, K. Okada and A. Srinivasan),
pp. 299-318, Springer-Verlag, Tokyo.
López-Bucio
J,, de la Vega O.M., Guevara-García A. and Herrera-Estrella L (2000)
Enhanced phosphorus uptake in transgenic tobacco plants that overproduce
citrate. Nature Biotechnology 18, 450-453.
Lugtenberg B.J.J.,
Dekkers L., and Bloemberg G.V. (2001)
Molecular determinants of rhizosphere colonization by Pseudomonas. Annual Reviews of Phytopathology 39, 461-490.
Macklon A.E.S,
Grayston S.J., Shand C.A., Sim A., Sellars S. and Ord B.G. (1997) Uptake and transport of phosphorus by Agrostis
capillaris seedlings from rapidly hydrolysed organic sources extracted from 32P-labelled
bacterial cultures. Plant and Soil 190, 163-167.
McLaughlin M.J.,
Alston A.M. and Martin J.K. (1988) Phosphorus cycling in wheat-pasture
rotations. II. The role of the microbial biomass in phosphorus cycling. Australian
Journal of Soil Research 26, 333-342.
Oberson, A.,
Friesen D.K., Rao I.M., Bühler S. and Frossard E. (2001) Phosphorus transformations in an oxisol
under contrasting land-use systems: The role of the microbial biomass. Plant and Soil 237, 197-210.
Oehl F., Oberson
A., Probst M., Fliessbach A., Roth H.R. and Frossard E. (2001) Kinetics of microbial phosphorus uptake
in cultivated soils. Biology
and Fertility of Soils 34, 32-41.
Raghothama K.G.
(1999) Phosphate acquisition. Annual Reviews of Plant Physiology and Plant
Molecular Biology 50, 665-693.
Randall P.J., Hayes J.E., Hocking P.J. and
Richardson A.E. (2001) Root exudates in phosphorus acquisition by plants. In Plant
nutrient acquisition: New perspectives (Eds N. Ae, J. Arihara, K. Okada and A. Srinivasan),
pp.71-100, Springer-Verlag, Tokyo.
Ranjard L., Poly F.
and Nazaret S. (2000) Monitoring complex bacterial communities using
culture-independent molecular techniques: Application to soil environment. Research
in Microbiology 151, 167-177.
Richardson A.E. (2001) Prospects for using
soil microorganisms to improve the acquisition of phosphorus by plants. Australian
Journal of Plant Physiology
28, 897-906.
Richardson A.E., Hadobas P.A., Hayes J.E.,
O’Hara C.P. and Simpson R.J. (2001a) Utilization of phosphorus by pasture plants supplied with myo-inositol hexaphosphate is enhanced by the
presence of soil microorganisms. Plant
and Soil 229, 47-56.
Richardson A.E., Hadobas
P.A. and Hayes J.E. (2001b) Extracellular secretion of Aspergillus phytase from Arabidopsis roots enables
plants to obtain phosphorus from phytate. The Plant Journal 25, 641-649.
Rodríguez H. and Frago R. (1999)
Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnology
Advances 17, 319-339.
Theron J. and
Cloete T.E. (2000) Molecular techniques for determining microbial diversity and
community structure in natural environments. Critical Reviews in
Microbiology 26, 37-57.
Thompson J.P.
(1994) What is the potential for management of mycorrhizas in agriculture? In Management
of mycorrhizas in agriculture, horticulture and forestry. (Eds A.D. Robson,
L.K. Abbott and N. Malajczuk) pp. 191-200. Kluwer Academic Publishers, Dordrecht.
Tinker P.B. (1980)
The role of rhizosphere microorganisms in mediating phosphorus uptake by
plants. In The role of
phosphorus in agriculture. (Eds. F.E. Kwasenah, E.C. Sample and E.J. Kamprath).
pp 617-654, American Society of Agronomy, Madison.
Tunney H., Carton
O.T., Brookes P.C. and Johnston A.E. (1997) Phosphorus loss from soil to water. CAB International, Oxon.
Whitelaw M. (2000)
Growth promotion of plants inoculated with phosphate-solubilizing fungi. Advances
in Agronomy 69, 99-151.